Gut Function-Enhancing Properties and Metabolic Effects of Dietary Indigestible Sugars in Rodents and Rabbits
Contents
Abstract
Indigestible sugars (iS) have received particular interest in food and nutrition research due to their prebiotic properties and other health benefits in humans and animals. The main aim of this review article is to summarize the current knowledge regarding digestive and health-enhancing properties of iS such as sugar alcohols, oligosacharides, and polysaccharides, in rodents and rabbits. Besides ameliorating gut health, iS ingestion also elicits laxative effects and stimulate intestinal permeability and fluid secretions, thereby shortening digesta transit time and increasing stool mass and quality. In rodents and rabbits, as hindgut fermenters, consumption of iS leads to an improved nutrient digestibility, too. Cecal fermentation of iS reduces luminal pH and extends wall tissue facilitating absorption of key dietary minerals across hindgut. The microbial fermentation of iS also enhances excessive blood nitrogen (N) flowing into the cecum to be used as N source for bacterial growth, enhancing N retention in cecotrophic animals. This review also highlights the impact of iS on improving lipid metabolism, mainly by lowering cholesterol and triglycerides levels in the blood. The paper serves as an index of the current knowledge of iS effects in rodents and rabbits and also identifies gaps of knowledge that need to be addressed by future research.
Introduction
Indigestible sugars (iS) include a large assembly of simple and complex carbohydrates including monosaccharides, disaccharides, oligosaccharides, and most sugar alcohols which have in common their crystalline nature, solubility in water, and, most importantly, resistance to host enzyme hydrolytic activity in the small intestine in humans and monogastric animals [1,2,3,4]. Due to their indigestibility, iS pass through the upper gastrointestinal tract and become readily available for microbes in the hindgut where they are degraded through various metabolic pathways to short-chain fatty acids (SCFAs) and gases. These degradation processes occur in the lumen and on the interface between the lumen and the gut epithelium [5]. In being water-soluble, microbial degradation metabolites can pass the intestinal barrier either through active absorption processes or facilitated diffusion. Both the chemical structure of iS and the glycosidic linkages between their molecules have a great impact on the site where the different iS are fermented and how the iS influence the physiology of the gastrointestinal tract [2]. In addition, due to the indigestibility and slow active transport across the small intestinal mucosa iS can increase the amount of osmotic active molecules in the intestinal lumen which, in turn, lead to an increased luminal water influx and thus contribute to the laxative properties of iS [3].
The rising popularity of so-called bioactive iS including, among others, disaccharide difructose anhydride (DFA) III and various oligosaccharides, such as fructooligosaccharides (FOS), xylooligosaccharides (XOS), mannanoligosaccharides (MOS), and galactooligosaccharides (GOS), have accelerated food science research in this area over the past two decades. Apart from their implication as low-calorie sweeteners and humectants, indigestible polyols (sugar alcohols), which are chemically defined as saccharide derivatives in which a ketone or aldehyde group is replaced by a hydroxyl group [6], such as maltitol, mannitol, sorbitol, erythritol and xylitol, have also been evaluated for their gastrointestinal health enhancing properties. Owing to their promising physiological effects, the interest in fortificating diets with iS has greatly increased and a large body of evidence in humans and animals has documented beneficial effects of using the various iS to ameliorate gut function and health as well as nutrient digestion and metabolic health [7,8,9].
The most extensively used animal models to examine physiological effects of iS include rats, mice, hamster, guinea pigs, and rabbits [10,11,12,13,14], and to a lesser extent the pig [15]. Rabbits, guinea-pigs and hamsters are small herbivores, whereas rats, mice and pigs are omnivores. Compared with large herbivores, such as ruminants and horses, these small herbivores have a lesser ability to break down and utilize dietary fiber, but they require more energy and protein per unit body mass than larger herbivores [16]. As hindgut fermenters, rodents and rabbits comprise either a large cecum or colon, where digesta is retained, mixed and fermented by microbes [16]. Rabbits have a large and sacculated cecum, which contain 40% of gastrointestinal digesta [17]. However, compared to rabbits, guinea pigs possess a larger cecum, containing 65% of gastrointestinal digesta and longer colon [17]. The colon in guinea pigs is capacious and never empty, which have a stronger ability to digest dietary fiber [18]. The hindgut of hamsters is lighter and shorter than that of rabbits and guinea pigs, but it is larger than in rats [19]. Chiou et al. [19] and Yu et al. [20] compared digestive function among rabbits, guinea pigs, rats and hamsters (Table 1). Guinea pigs showed highest crude fiber digestibility due likely to their greatest relative weight and longest length of colon-rectum, compared with other animals. Fiber-hydrolytic activity in the cecum of guinea pig was highest, too [20]. In contrast, rats showed the lowest fiber hydrolytic activity in the cecum and smallest relative weight of the hindgut, which corresponded to the lowest capability of the latter animals to digest fiber (Table 1). Rabbits possess the longest retention time of digesta in the gastrointestinal tract, followed by guinea pigs, hamsters and rats, which was the shortest [19]. The passage rate of digesta through the hindgut seems to be related to the relative length and the degree of sacculation of the local section [21]. In addition, the short dense and folded mucosa appears primarily in rabbits, and followed by guinea pigs [19]. Thus, compared with rats, hamsters and mice, rabbits and guinea pigs may provide larger volume for iS fermentation and production and absorption of SCFA. The total bacteria concentration and pH in the hindgut in rabbits, guinea pigs, rats and mice are similar [21]. However, the species, population and distribution of bacteria in rabbits are closer to the colonic microflora in humans [21]. Indeed, similarities and major differences in the gut physiology and microbiota between rodent models and humans have been recently reviewed [22,23]. When rodents and rabbits are used as animal models to study the effects of iS in human gut, metabolic limitations exist due to the anatomical, physiological and microbial differences between these animals and humans [21,22,23]. For example, differing from rodents and rabbits, the cecum is a small dead-end pouch forming, and has less bacterial activity in humans [21]. In contrast, humans have longer and more sacculated colon which consists of four sections called the ascending colon, the transverse colon, the descending colon and sigmoid colon [22]. Human colon contains larger population of bacteria in the gastrointestinal tract than the rodents and rabbits do [21,22]. Therefore, after the ingestion of iS, the colon is responsible for iS fermentation in humans. The colon also is the major site of production and absorption of SCFA, in addition, absorption of water, salts and minerals in humans.
In this article, we summarize beneficial effects of non-digestible monosaccharides, disaccharides, oligosaccharides and sugar alcohols including prebiotic effects and the related desirable physiological changes in the large intestine after oral ingestion of various types of iS as well as their putative positive health benefits on nitrogen, mineral and lipid metabolism using results originating mainly from rodents and rabbit research. Despite differences mentioned above, rodents are often used as gut and metabolic models in humans; therefore, a few examples of beneficial effects of iS in this article also come from human studies.
Prebiotic Properties of iS and Effects of Fermentation
Well-known nutrition-based strategies to modulate the gut microbiota and sustain gut homeostasis are based on the use of dietary fiber and analogous carbohydrates [24]. Due to many benefits of iS in the prevention and control of chronic human diseases and the fact that many of these effects are mediated via stimulation of fermentation in the large intestine, there is ongoing research interest in evaluating the potential of iS to be used in diet preparations as prebiotic [25]. A prebiotic is generally defined as a selectively fermented dietary ingredient that confers a health benefit on the host in association with modulation of the intestinal microbiota or microbial activity [26]. Various known prebiotic effects of iS in rodent and rabbit models are discussed in this section and are summarized in Figure 1.
Selective Bacterial Proliferation and Fermentation
The main end products of the fermentation of iS are SCFAs, predominantly acetic acid, propionic acid and butyric acid, and lactic acid, as well as water, various gases (carbon dioxide, hydrogen, methane), bacterial cell biomass [27,28] and heat. The production of SCFAs is influenced by several factors, including the number and microbes present in the large intestine [26], type and mass of substrate [29], and gut transit time [30]. Thus, different types of iS lead to diverging SCFAs patterns in the hindgut. For instance, FOS increased mainly butyric acid and acetic acid concentrations in the human colon [31]. Mannitol, in turn, has showed to stimulate the generation of lactic acid, succinic acid and butyric acid and to reduce that of acetic acid in the hindgut and feces of rats, whereas inconsistent reports exists regarding propionate fermentation [32,33]. Difructose anhydride III feeding to rats increased especially acetic acid formation [34] which could be linked to the enhanced abundance of Ruminococcus sp. M-1 withdietary DFA III.
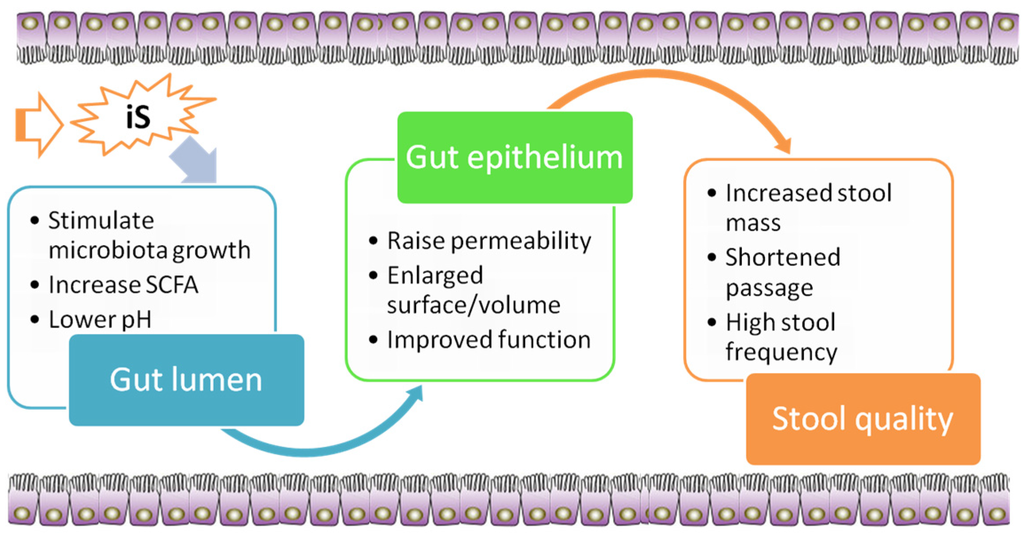
Traditionally, the main health benefits of iS consumption on the intestinal microbiota have been related to selective promotion of saccharolytic bifidobacteria and lactobacilli species, while harmful bacteria such as Enterobacteriaceae, Clostridium sensu strictu, Streptococcus faecalis and Proteus sp. typically decreased following iS intake in human large intestine [35,36]. For instance, FOS intake induced a significant increase in the population of Lactobacillus spp. and Bifidobaterium spp., which came along with increased levels of butyric acid and acetic acid in human colon in vitro [31]. Of particular interest is the promotion of butyric acid-producing bacteria (e.g., Faecalibacterium prausnitzii and Eubacterium rectale/Roseburia spp.) through iS due to the beneficial effects of butyric acid on colonic health, as reported in humans [37] and rats [38]. Moreover, iS effect on microbiota in the different intestinal segments differs, which can be related to progressing fermentation of iS. In this regard, FOS increased fecal gene copy number of Bifidobacterium spp., and mediated a decrease in gene copies of total bacteria, Bacteroides-Prevotella-Porphyromonas group, and Clostridium clusters XI and XIVa, Enterobacteriaceae and Clostridium difficile toxin B in feces of rats [38]. In the cecum, in turn, FOS increased Bifidobacterium spp. and Clostridium cluster XI [38].
Apart from the direct effect of substrate preferences of bacteria and competition for carbohydrates among bacteria, the drop in large intestinal pH due to enhanced fermentation of iS inhibits the growth of acid-sensitive bacteria including some intestinal bacterial pathogens such as Enterobacteriacae [37], Salmonella [39] and Clostridium species [40,41], Escherichia coli [42] and provides a growth-advantage of more acid-resistant bacteria, including lactobacilli and bifidobacteria that withstand lower environmental pH [43,44,45,46].
Moreover, Lactobacillus and Bifidobacterium species are well recognized to suppress opportunistic intestinal pathogens such as Bacteroides, Salmonella enteric, Clostridium diffile, Staphylococcus, and Enterobacter in gastrointestinal tract via production of antimicrobial metabolites [44,47,48]. Stimulation of lactobacilli and bifidobacteria species by iS may therefore contribute to normal gut function and maintaining host health in a number of ways. GOS supplementation at 2% (w/v) was the optimum dosage for growth of Lactobacillus acidophilus NCDC13 after 12 h culture in vitro [49]. In rats, dietary GOS was observed to increase lactobacilli counts and decrease coliform counts in feces [50]. In addition, dietary DFA III decreased the population of bacteria related to Bacteroides acidofaciens and uncultured bacteria within the Clostridium lituseburense group and increased the population of bacteria related to Bacteroides vulgatus, Bacteroides uniformis and Ruminococcusproductus in the rat cecum [34].
Effects of Microbial Metabolites on the Gut
Short-chain fatty acids are an important nutrient source of the gut epithelium [51]. Especially butyrate stimulates cell proliferation of the cecal and colonic mucosa in humans and animals like pigs and rats [52,53,54,55]. After first pass uptake, the remaining butyrate and propionate are metabolized by hepatocytes. In addition, 50%–70% of acetate is taken up by the liver, thereby contributing to energy intake of the host. Roberfroid [26] reported that muscle cells generate energy from the oxidation of residual acetic acid. Increased luminal generation of SCFAs in the large intestine has been related to epithelial cell proliferation after the ingestion of iS [56]. For instance, dietary MOS supplementation has been shown to enhance cecal SCFAs concentrations in rabbits which was associated with longer intestinal villi and an increased intestinal absorptive surface [57,58].
Emerging evidences from human researches also suggest positive effects of certain SCFAs on gut mucosal immunity and attenuation of the inflammation in the large intestine. For example, butyrate can be protective against carcinogenesis [59], or act anti-inflammatory in colon cancer cells [60]. In addition, newer research has indicated that butyrate regulates gut mucosal homeostasis and elicits modulatory effects on dendritic cells [61] and on the differentiation of colonic regulatory T cell [62,63,64]. A new study in rats also showed that a short term (1 week and 2 weeks) ingestion of FOS significantly increased IgA and mucin concentrations in cecum, but this effect was attenuated with a prolonged ingestion [65], indicating development of a state of tolerance due to long exposure of this dietary treatment. Indeed, most of the studies dealing with immune-stimulating effects of iS ingestion are done in short-term studies. More long term designed studies are warranted to clarify the role of iS ingestion in modulation of mucosal immunity.
Enlargement of Large Bowel Size
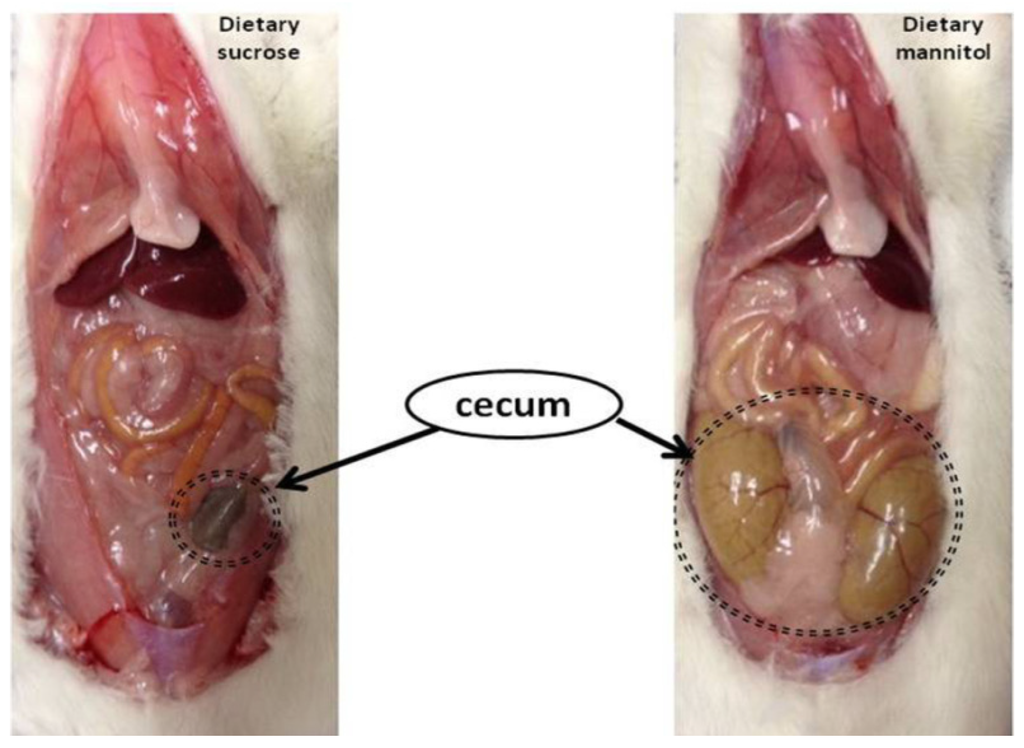
An enlargement of intestinal bowel has also been recognized as one beneficial physiological effect of iS ingestion. Important implications of enlargement in large bowel size are greater fermentation capacity and an increasing role of the hindgut in nutrient absorption in those animals. In rats, greater substrate availability for fermentation in the large intestine, due to mannitol supplementation, led to an enlargement of cecum compared to rats consuming digestible sugars such as sucrose (Figure 2). Indeed, in this research, as shown in Figure 2, dietary mannitol supplementation resulted in multifold larger and heavier cecum than in rats fed dietary sucrose.
Bigger hindgut in response to iS feeding is a typical finding in rodent studies [9,12,66,67,68,69]. For example, research conducted by Pan et al. [68] revealed that daily gavage of FOS, GOS, MOS, and chitooligosaccharides (delivered in a quantity of 1 g/kg body weight) to the mice for two weeks led a significant increase in the total weights of cecum and colon, associated with a reduced cecal pH. The cecal SCFAs and total anaerobes in the cecum were increased by these 4 oligosaccharide supplementations as well. When FOS was fed to rats, an enlargement of cecum and a decrease in cecal pH was reported, too [69]. In hamsters, 16% inulin feeding increased total cecal weight and cecal wall weight as well [12].
Change in Intestinal Transit Time and Increase Stool Mass
Indigestible sugar supplementation in the diet has been shown to increase stool mass and shorten transit time of digesta through gastrointestinal tract. For example, the gut transit time was significantly reduced with 6% FOS supplemetation in rats [66]. The increased stool mass has mainly been associated with higher amounts fecal water and greater amounts of bacterial biomass [35]. In addition, the fermentation of iS is believed to result in greater production of microbial biomass in large intestine, increasing this biomass in feces. Indeed, the bacterial biomass accounts for at least 30% on dry matter of feces [35], which is responsible for the increased fecal bulk and dry matter excretion. Daily ingestion of GOS (3.5 and 7 g/day) enhanced fecal biomass of Bifidobacteria in patients suffering from irritable bowel syndrome [71].
The role of iS consumption on stool weight and size has also be related to modulation of the transit time by iS fermentation in the large intestine. Particularly butyric acid seems to impact the transit time through the gastrointestinal tract, as greater proportions of butyric acid lead to a shorter cecal transit time [30]. Oral ingestion of FOS effectively increased stool frequency, shortened the transit time of digesta and increased fecal weight in mice [72]. In human studies, the ingestion of FOS significantly increased the bowel evacuation rate, reduced perception of straining effort and pain, and improved the quality of stools [72]. A specific mixture of GOS and long-chain FOS enhanced stool frequency, led to softer stools [73], and accelerated gastrointestinal transport in infants, too [74]. In addition, XOS intake increased the population of intestinal Bifidobacterium in humans, maintained normal fecal water content, and reduced constipation [75]. Therefore, iS could be considered as useful and safe tool for ameliorating constipation.
Indigestible sugars alter the intestinal permeability in two ways: (1) through their physical presence, and (2) through fermentation. Physical presence affects several physiological functions in the upper intestine. Indigestible sugars produce high osmotic pressure, accumulate fluid within the lumen to maintain isotonicity, and increase the permeability of the intercellular junctions in small intestine in rats [76]. On the other hand, excessive consumption of iS may induce osmotic diarrhea [77]. In large intestine, osmotic pressure is caused by the high production of SCFAs during the fermentation of iS. The transport process of SCFAs is accompanied by increased Na, K, and water absorption [78]. An increased fluid secretion through a greater luminal osmotic pressure results in softer stool. Therefore, osmotic effect of iS can be considered as one of the reasons for their laxative effects besides increasing the bacterial biomass.
Effects on Nutrient Digestion, Absorption, and Systemic Metabolism
Enhancement of Mineral Absorption
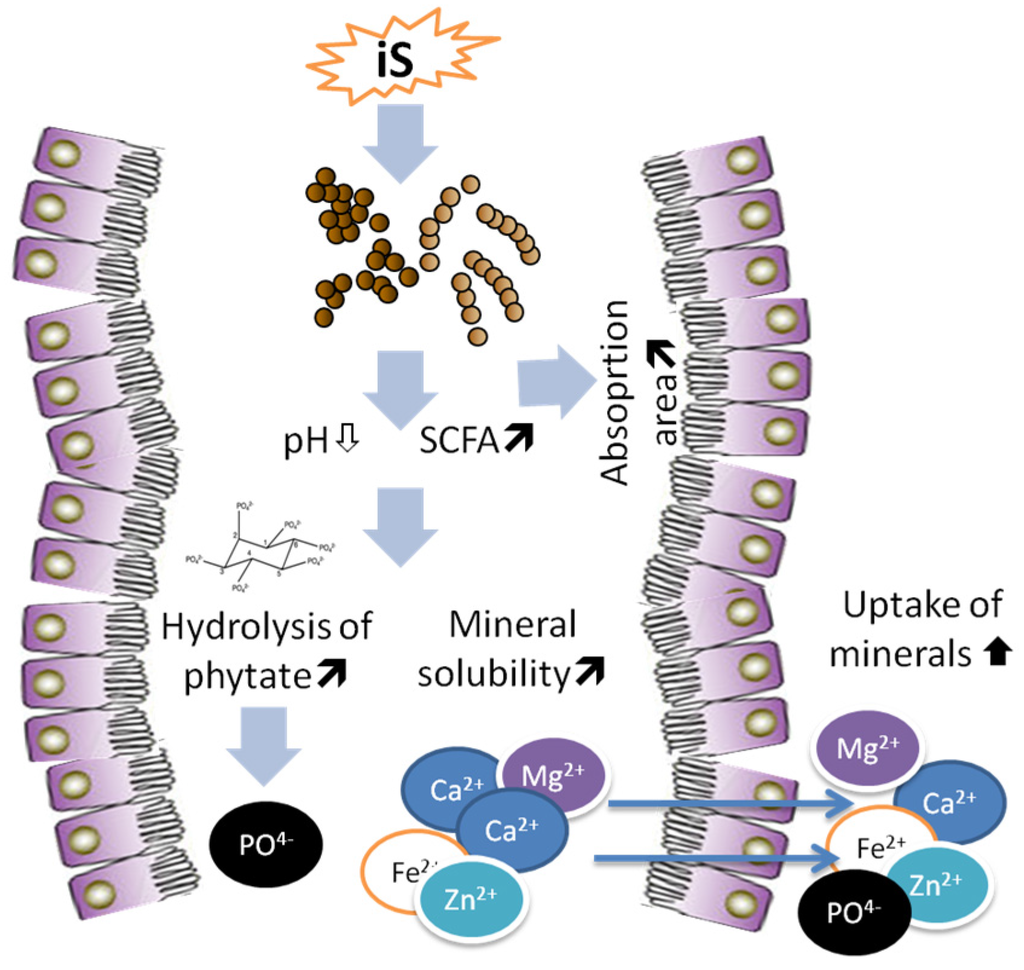
Improvement of mineral bioavailability is one important properties of iS, which when fermented in large intestine stimulate microbial activity, SCFAs formation, and eventually an enlargement of cecum, thus playing an important role in mineral absorption (Figure 3). Indeed, increasing evidences suggest that iS supplementation promoted mineral absorption including Ca, P, Mg, Zn, and Fe with key physiological functions in the body. Most of the scientific evidences of the effects of iS is based on the results of experiments with rats, whereby iS increased the availability of Ca, Mg, Fe and Zn [71,79,80,81,82,83], which are the most important mineral elements for bone mineralization and bone health [84]. The ingestion of lactosucrose, which is synthesized from sucrose and lactose, increased the apparent Ca absorption and Ca accumulation in the bone of rats [85]. Supplementation of FOS in the diet enhanced the mineral absorption, and, most importantly, counteracted the adverse effects of phytic acid in mice [69]. This is highly important because dietary P is mostly in form of phytic acid in cereal- and legume-rich diets, which is largely unavailibale for the host animal [86]. Thus, conteraction of phytic acid by iS supplementation [56] increases the availability of dietary P [86].
Other research also found that dietary GOS enhanced net absorption of Ca and Mg, as well as their retention, and femur and tibia breaking strength in rats [87]. Both the distal femur, trabecular volumetric bone mineral density, and proximal tibia volumetric bone mineral density increased with GOS supplementation, too [87]. Feeding of GOS for 9 days to growing rats with hypochlorhydria significantly improved apparent absorption of Ca, Mg and Fe [82,88]. In particular, the effect was stronger when GOS ingestion was combined with a dairy product fermented by Lactobacilli, resulting in improved Ca, P, Fe and Zn retention during the mineral balance test in rats [88]. Ingestion of FOS has also been proven to improve intestinal absorption of Ca, Mg, Fe, Zn, and Cu in rats [54,89,90,91]. Ingestion of indigestible polyols increased Ca content in bone [92,93,94,95] and bone strength [96,97] in rats. Dietary mannitol has been shown to increase Ca and Mg absorption and their retention in the bone of rats, too [33].
Typically, beneficial effects of iS on enhancing the bioavailability of minerals were combined with improvements of SCFAs formation, decreased luminal pH, and enlargement of intestinal wall tissue in the above researches. Hydrolysis of phytic acid contained in the cereal-based diets via iS fermentation [69] may also contribute to a higher bioavilibility of P and other minerals such as Ca, Mg, Zn, which normally are bound by phytic acid [86,89].
Following the ingestion iS, the absorption of minerals is shifted towards the large intestine in rats [98]. Dietary DFA III increased Fe absorption through expansion of cecal mucosa [99]. Cecum is the main segment with highest Ca absorption in the rat intestine, with active transport of Ca in cecum being several times faster than in duodenum, proximal colon and distal colon in rats [100]. Under normal conditions, cecal epithelium absorbs free-ionized minerals released from insoluble mineral complexes in the presence of small acidic molecules, such as acetic, propionic, butyric, succinic and lactic acids which are formed during the fermentation of iS by luminal microbes [98,101]. Indigestible sugars fully or partly reach the cecum and are fermented there, producing SCFAs which are responsible for the drop of cecal pH in rats (Figure 3). It was shown that inulin and FOS improved mineral absorption and the effects were associated with SCFAs formation and reduced luminal pH [102]. The drop of cecal pH increases the concentration of mineral cations [103,104]. Raschka and Daniel [105] reported that a lower pH increased the amount of soluble and ionized minerals, which was adequate for their absorption.
Enlargement of cecum and colon induced by iS consumption and the resulting SCFAs provide extensive absorption area to stimulate mineral absorption in trials involving piglets, rats, and chicken [33,52,69,106,107]. Subsequently, the SCFAs may directly affect intestinal mineral absorption by stimulating the intestinal epithelium and increasing its absorptive capacity and by increasing intestinal blood flow and fluid and electrolyte uptake, as reported in human colon [108]. Short chain fatty acids, especially butyrate formed by iS fermentation, increase the energy supply to intestinal epithelial cells and regulate the electrolyte exchange of minerals and hydrogen to facilitate mineral absorption by the epithelial cells in the rat colon [109].
Moreover, iS ingestion increases bone mineral accumulation, where by this effect depends on the dose and type of these sugars and mineral level in diet of rats [110]. The benefit function is most effective when high levels of minerals in the diet are supplemented in rats [101]. Feeding Fe-deficient rats with GOS together with FeCl3 increased hemoglobin (Hb) synthesis (by 17%) and mean Hb concentration in the erythrocytes relative to FeCl3 level in the diet [13]. Dietary heme iron did not prevent postgastretomy anemia alone, but dietary FOS increased iron absorption and prevented anemia when its consumption was combined with iron citrate or heme iron in gastrectomized rats [111].
In these years, the major part of researches on iS has been designed in mineral deficient animals or animals with mineral malabsorption. Ohta and colleagues confirmed that dietary FOS enhanced Mg absorption and the absorption of Ca, Mg and Fe in Mg-deficient rats and Fe-deficient anemic rats respectively [112,113]. As well, dietary supplements of GOS, FOS, DFA III, polydextrose and epilactose were reported to increase Ca and Fe bioavailability and prevent anemia in gastrectomized animal models [111,114,115,116,117].
Ovariectomy has been well known to induce bone mineral loss and osteopenia. Mineral absorption such as Ca, Mg and Fe was increased and bone mineral loss was prevented by dietary FOS, GOS, DFA III, maltitol and polydextrose in ovariectomized rats [106,110,118,119]. Pieces of evidence suggest that Ca retention was greater after 11 day supplementation of iS, but no longer after 25 days in rats [120]. Therefore, the effects of iS on mineral absorption and retention may be related to the length of feeding period too. Because most of the studies are conducted during short periods of time, elucidation of this aspect requires further research with long-term designed studies.
An increasing body of evidence suggests that iS enhances mineral absorption across small intestine wall, too. For instance, dietary lactose induced stimulation of Mg absorption in rats, and this was caused by a lowering effect on ileal pH [121]. Using a tracer technique with 45Ca, maltitol was found to stimulate Ca absorption in rats, and osmotic activity of maltitol in small intestine was suggested to contribute to the increase in Ca absorption [122]. Other iS such as melibiose, DFA III, or DFA IV increased the permeability of intercellular passage, affected the epithelial tissue and opened the tight junctions of jejunum, ileum, cecum, and colon of rats [123]. Absorption of Ca, Mg, and Zn via the paracellular route was enhanced, thereby promoting Ca, Mg, and Zn absorption in the small intestine and large intestine in vitro [123,124]. In these in vitro studies, the stimulating effect of iS on mineral absorption was related to greater epithelial permeability, opening the tight junctions to facilitate mineral absorption in small intestine.
In a small number of studies, the researchers found that iS supplementation also have similar promoting effects on mineral bioavailability in rabbits and other rodents except of rats. Dietary mannitol increased ash digestibility in adult rabbits [125,126]. A research by Kawasaki et al. [10] showed that dietary FOS resulted in significantly higher ash digestibility in guinea pigs when compared with dietary glucose.
Nitrogen Utilization
Fermentation of iS in the hindgut serves as the source of energy and carbons for bacterial growth leading to a distinguished rise of bacterial biomass. Besides available energy, dietary N is also needed for bacterial proliferation, whereby bacteria use ammonia N for their protein synthesis. Thus, the higher the availability of energy and fermentation substrates (i.e., iS sources), the greater is the need of the hindgut bacteria for ammonia N, needed for their rapid growth. In rats, supplementation of dietary mannitol at 8% lowered the digestibility of crude protein compared with dietary sucrose [127]. In addition, in rats, feeding diets containing GOS or FOS at 100 g/kg decreased the apparent digestibility of crude protein when compared with the diet including the same dose of sucrose [128]. During iS supplementation period, the fermentation of these substrates resulted in a greater flux of urea N transfer from blood into cecum to provide N for cecal bacterial proliferation. Dietary FOS and XOS increased N cecal flux from blood urea N into cecum too, associated with greater cecal N excreted in feces as bacterial N [129]. This circulation of N can explain the decreased crude protein digestibility in rats when they fed dietary sources of iS. Therefore, more urea N is excreted in feces when bacterial mass increased due to iS fermentation, coupled with a reduction in urinary N excretion and plasma urea concentration. For example, when FOS and XOS were fed at 7.5 g/100 g in rats, blood urea and urinary N were reduced by 20%–30% [129].
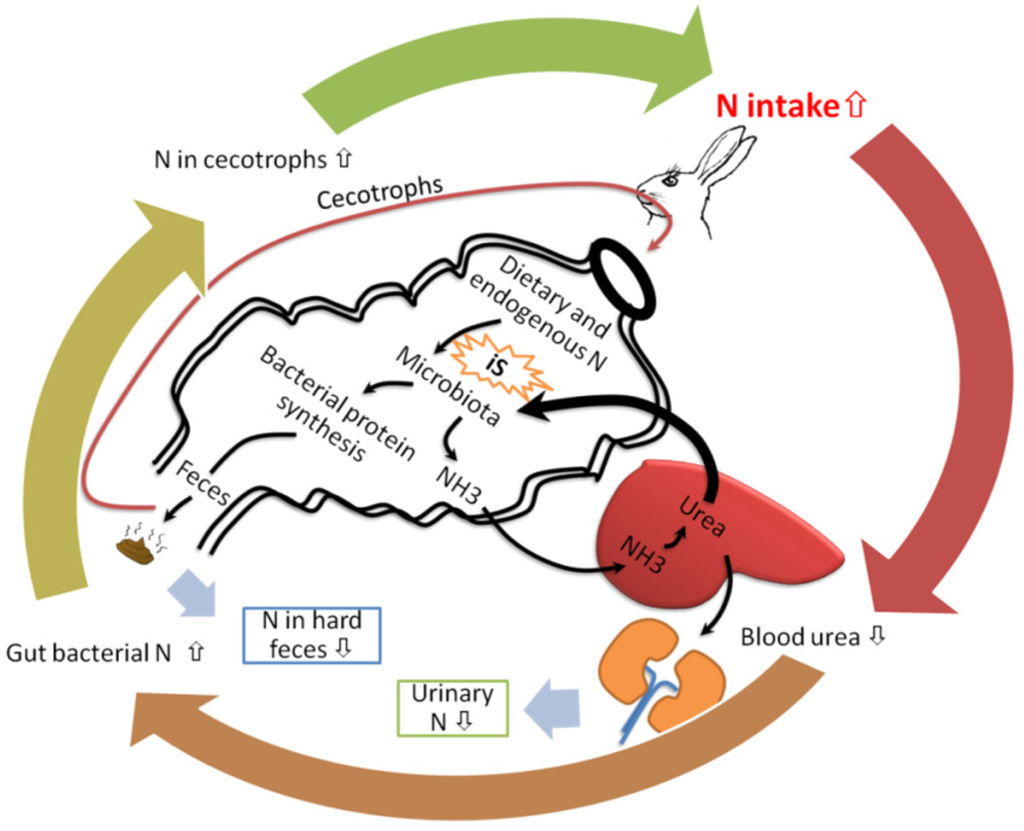
The greater amount of bacterial biomass in fecal dry matter plays an important role in cecotrophic animals like rabbits and guinea pigs. Because rabbits and guinea pigs are typical cecum fermenters, and have a special reingestion system called cecotrophy which happens less in rats. Rats also eat their feces, but this action only happens when rats are under the condition of starving. Cecotrophy allows rabbits and guinea pigs to reingest their microbial product in the cecum, called cecotrophs or soft feces. Fecal N source includes the part of dietary protein escaped from digestion in the small intestine, endogenous proteins secreted from pancreas and intestine, mucosa cells sloughed from intestinal wall, blood urea that diffuses across the intestine with water movement, and intestinal bacteria. Bacterial protein is the major part of cecotrophs N [130]. Cecotrophs is formed in the cecum without any major differences in their composition when it is excreted [131]. Li et al. [125] revealed that crude protein concentration in cecotrophs was raised by iS feeding when rabbits were prevented from cecotrophy, and N excretion in urine was simultaneously reduced. The raised N in cecotrophs results from bacterial proliferation which is stimulated by iS in the cecum. The fermentation of iS induces excessive amounts of blood urea flowing into the cecum [132], which is transformed to ammonia by bacterial ureolytic activity [133] to contribute to bacterial N synthesis [128,134]. As a result, urinary excretion of N is shifted to fecal excretion [10,128]. The cecotrophs with a rich bacterial N are eaten by rabbits and guinea pigs, and bacterial N is absorbed in small intestine to increase N retention [135]. Rabbits fed 1 g/kg dietary MOS had higher overall final live weight, weight gain among the diets containing several probiotics and prebiotics [14]. In asimilar research in guinea pig, it was shown that 5% dietary FOS depressed urinary N excretion, and increased N retention [10]. The increased N retention caused by dietary iS supplementation in rabbits and guinea pigs were attributed to cecotrophy in these animals [10,14].
The improved utilization of dietary N in rabbits and guinea pigs by iS intake is summarized in Figure 4. Nitrogen from the diet in rabbits and guinea pig is metabolized in a circle, a physiological process called N recycling [134]. Indigestible sugars stimulate blood urea move to cecum to be used as N source for bacterial proliferation, raising bacterial protein in soft feces [136]. Soft feces are reingested through cecotrophy. Cecotrophy has considerable nutritional significance for N supplementation in rabbits. It can provide up to 30% of daily N intake, which is mostly derived from cecal microbes. The utilization of dietary urea is more effective to increase N retention in rabbits when iS are supplemented in the diet [136]. Therefore, the positive effects of iS in cecotrophs N can make a significant contribution to meeting the N requirements in cecotrophic animals (Figure 4).
Role in Improving Blood Glucose and Insulin Secretion
Indigestible sugars are not directly converted into energy because of the resistance to be digested and utilized in the small intestine. Therefore, they do not stimulate an increase in blood glucose and insulin secretion. Oral administration of FOS did not change the level of glucose, fructose, and insulin in plasma, indicating that FOS was not absorbed directly into blood both in rats and humans [137]. In addition, the consumption of FOS did not result in changes of serum lipid profiles and glucose levels, but led to 3.6 fold less serum insulin concentrations compared to sucrose diet in rats [138]. FOS in the diet caused a decrease in serum glucose after 14 days addition in subjects with diabetes type II, but this effect was not shown when FOS was supplied in healthy subjects [139]. Blood glucose, total serum cholesterol and liver ascorbic acid were reduced by the ingestion of galactitol, mannitol, as well as xylitol in rats, too [140]. Sangwan et al. [50] found that dietary GOS decreased diabetes-associated markers including fasting blood glucose, haemoglobin, glycosylated haemoglobin triglycerides, total cholesterol, low density lipoproteins, creatinine, and urea in alloxan-induced diabetic rats. In human research, matitol and short chain FOS containing food resulted lower blood glycaemic and insulinaemic responses than the food with traditional sugars [141]. Plasma glucagon level was decreased by dietary sorbitol and dietary xylitol, but plasma insulin and glucose was little affected by the addition of these iS sources in rat studies [142].
Lipid Metabolism
The ingestion of iS decreased feed efficiency and body weight gain in rats, which has been mainly attributed to a decreased fat digestibility [127]. The latter research showed that dietary mannitol decreased the level of serum triglycerides and fat accumulation in body in a dose-response manner [127]. A similar result was shown with dietary sorbitol and xylitol which decreased plasma triglyceride and cholesterol, and fat tissue weights in rats [142]. Matitol supplementation suppressed weight gain, hepatic fat degeneration, hyperglycemia and hypercholesterolemia in mice fed high-fat diets [143] In addition, the epilactose in the diet lowered the levels of plasma total cholesterol and low-density lipoprotein cholesterol in rats [144]. Dietary FOS also showed a hypolipidemic effect in rats, for example, decreasing total cholesterol and triglycerides in blood [145]. In addition, a 12-week intragastrical administration of FOS reduced body weight, blood cholesterol and triglycerides in rats with fructose-induced obesity [146]. Dietary inulin lowered plasma cholesterol and triacylglycerol in hamsters [12], and the related mechanisms were postulated that inulin ingestion altered hepatic triacylglycerol synthesis and very-low-density lipoprotein (VLDL) secretion and impaired rebsorption of circulating bile acids. Bile acids excretion in feces was increased by inulin ingestion [12]. Cholesterol is used to synthesize new bile acids in the liver to compensate the loss of bile acids, resulting in lowered levels of serum cholesterol [147]. Taylor and Williams [148] demonstrated that the consumption of FOS resulted in marked reductions in triglycerides, and to a lesser extent cholesterol levels, because FOS suppressed hepatic triglyceride and VLDL synthesis. Inulin and FOS are short- and long- chain, linear fructans, consisting of fructose monomers bound with b-2,1-glycoside bonds [149]. Fructans are not viscous and incapable to bind with bile acids in the intestine. Thus, the hypolipeamic effects of FOS and inulin are attributed to SCFAs (mainly acetate, propionate, and butyrate) formed during the fermentation of fructans in the large intestine, which may inhibit enzymes involved in triglyceride and cholesterol synthesis in the liver in humans and rats [150,151]. It has been proved that dietary inulin-type fructans have reducing effects on TG and cholesterol in rodent studies. In human, most studies support positive effects of fructans in lipid metabolism, but the conflicting results still exist, especially in healthy subjects [151]. In addition, the consumption of iS such as soybean oligosaccharides and chitooligosaccharides increased high-density lipoprotein (HDL)-cholesterol in rats [152] and chicken [153] studies. A decreased serum low-density lipoprotein (LDL)-cholesterol with increased serum HDL-cholesterol was reported in Sprague-Dawley hypercholesterolemic-induced rats fed inulin [154]. A similar result was observed whereby serum total lipids, cholesterol, and LDL-C and VLDL-C were decreased whereas HDL-C was increased with 5% MOS supplementation in hyperlipidemic albino rats [155]. High-density lipoprotein cholesterol is involved in the reverse cholesterol transport, scavenging and removing bad cholesterol from the body. Although the mechanism of increasing effect of iS on HDL-cholesterol is unclear, it can be another explanation for hypolipeamic effects of iS activity.
Conclusions
This review primarily covers gut function-enhancing properties of iS and their metabolic effects in rodents and rabbits. The majority of the studies reviewed herein suggest prebiotic effects of the ingestion of iS, which include improvement of the microenvironment in the hindgut and constipation, as well as ameloriation in the nutrient metabolism, especially mineral absorption in the hindgut. Future studies are needed to evaluate long-term impact of iS supplementation as low caloric food supplement on mineral bioavailability. In particular, more clinical results are necessary in special cases such as patients suffering from mineral malabsorption or bone mineral losses due to aging, obesity or diabetes. In addition, more long-term designed studies are warranted to establish the effects of a prolonged supplementation with iS. Due to their beneficial effects on enhancing N utilization and micronutrient absorption, the use of iS offers great potentials not only in improving gut health and animal production, but also in decreasing environmental pollution via less excretion of N and minerals from animals.
Author Contributions
Jin Xiao, Qendrim Zebeli designed and concepted the article, Jin Xiao, Barbara U. Metzler-Zebeli, Qendrim Zebeli retrieved and screened the articles for inclusion in the paper, Jin Xiao, Barbara U. Metzler-Zebeli, Qendrim Zebeli wrote and approved the article, Jin Xiao, Barbara U. Metzler-Zebeli, Qendrim Zebeli are responsible for the content of the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yanina Pepino, M. Metabolic effects of non-nutritive sweeteners. Physiol. Behav.2015. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Levin, G.V.; Zehner, L.R.; Saunders, J.P.; Beadle, J.R. Sugar substitutes: Their energy values, bulk characteristics, and potential health benefits. Am. J. Clin. Nutr.1995, 62, 1161–1168. [Google Scholar]
- Livesey, G. The energy values of dietary fiber and sugar alcohols for man. Nutr. Res. Rev.1992, 5, 61–84. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, K.E.B.; Lærke, H.N. Carbohydrate digestion and absorption. In Nutritional Physiology of Pigs; Bach Knudsen, K.E., Kjeldsen, N.J., Eds.; Videncenter for Svineproduktion: Foulum, Denmark, 2012. [Google Scholar]
- Zumbé, A.; Lee, A.; Story, D. Polyols in confectionery: The route to sugar-free, reduced sugar and reduced calorie confectionery. Br. J. Nutr.2001, 85, 31–45. [Google Scholar] [CrossRef]
- Sabater-Molina, M.; Larqué, E.; Torrella, F.; Zamora, S. Dietary fructooligosaccharides and potential benefits on health. J. Physiol. Biochem.2009, 65, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Tremaroli, V.; Bäckhed, F. Functional interactions between the gut microbiota and host metabolism. Nature2012, 489, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Grabitske, H.A.; Slavin, J.L. Gastrointestinal effects of low-digestible carbohydrates. Crit. Rev. Food. Sci. Nutr.2009, 49, 327–360. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, K.; Min, X.; Nishiayama, A.; Sakaguchi, E. Effect of fructo-oligosaccharide on nitrogen utilization in guinea pigs. Anim. Sci. J.2013, 84, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Bindels, L.B.; Neyrinck, A.M.; Salazar, N.; Taminiau, B.; Druart, C.; Muccioli, G.G. Non digestible oligosaccharides modulate the gut microbiota to control the development of leukemia and associated cachexia in mice. PLoS ONE2015. [Google Scholar] [CrossRef] [PubMed]
- Trautwein, E.A.; Rieckhoff, D.; Erbersdobler, H.F. Dietary inulin lowers plasma cholesterol and triacylglycerol and alters biliary bile acid profile in hamsters. J. Nutr.1998, 128, 1937–1943. [Google Scholar] [PubMed]
- Laparra, J.M.; Díez-Municio, M.; Herrerob, M.; Morenob, F.J. Structural differences of prebiotic oligosaccharides influence their capability to enhance iron absorption in deficient rats. Food Funct.2014, 5, 2430–2437. [Google Scholar] [CrossRef] [PubMed]
- Oso, A.O.; Idowu, O.M.O.; Haastrup, A.S.; Ajibade, A.J.; Olowonefa, K.O.; Aluko, A.O.; Ogunade, I.M.; Osho, S.O.; Bamgbose, A.M. Growth performance, apparent nutrient digestibility, caecal fermentation, ileal morphology and caecal microflora of growing rabbits fed diets containing probiotics and prebiotics. Livest. Sci.2013, 157, 184–190. [Google Scholar] [CrossRef]
- Halas, V.; Nochta, I. Mannan oligosaccharides in nursery pig nutrition and their potential mode of action. Animals2012, 2, 261–274. [Google Scholar] [CrossRef]
- Sakaguchi, E. Digestive strategies of small hindgut fermenters. Anim. Sci. J.2003, 74, 327–337. [Google Scholar] [CrossRef]
- Johnson-Delaney, C. Anatomy and physiology of the rabbit and rodent gastrointestinal system. Proc. Assoc. Avian Vet.2006, 9–17. [Google Scholar]
- Franz, R.; Kreuzer, M.; Hummel, J.; Hatt, J.M.; Clauss, M. Intake, selection, digesta retention, digestion and gut fill of two coprophageous species, rabbits (Oryctolagus cuniculus) and guinea pigs (Cavia porcellus), on a hay-only diet. J. Anim. Physiol. Anim. Nutr.2011, 95, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Chiou, P.W.S.; Yu, B.; Kuo, C.Y. Comparison of digestive function among rabbits, guinea pigs, rats and hamsters. I. Performance, digestibility and rate of digesta passage. Asian-Aus. J. Anim. Sci.2000, 13, 1499–1507. [Google Scholar] [CrossRef]
- Yu, B.; Chiou, P.W.S.; Kuo, C.Y. Comparison of digestive function among rabbits, guinea pigs, rats and hamsters. II. Digestive enzyme and hindgut fermentation. Asian-Aust. J. Anim. Sci.2000, 13, 1508–1513. [Google Scholar] [CrossRef]
- Kararli, T.T. Comparison of the GI anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm. Drug Dispos.1995, 16, 351–380. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.L.A.; Vieira-Silva, S.; Liston, A.; Raes, J. How informative is the mouse for human gut microbiota research? Dis. Mod. Mechan.2015, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.K.; Hartmann, C.; Hansen, F.; Krych, L.; Nielsen, D.S. Impact of the gut microbiota on rodent models of human disease. World J. Gastroenterol.2014, 20, 17727–17736. [Google Scholar] [PubMed]
- Lattimer, J.M.; Haub, M.D. Effects of dietary fiber and its components on metabolic health. Nutrients2010, 2, 1266–1289. [Google Scholar] [CrossRef] [PubMed]
- Zaman, S.A.; Sarbini, S.R. The potential of resistant starch as a prebiotic. Crit. Rev. Biotechnol.2015, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.B. Inulin-type fructans: Functional food ingredients. J. Nutr.2007, 137, 2493–2502. [Google Scholar]
- Gibson, G.R.; Fuller, R. Aspects of in vitro and in vivo research approaches directed toward identifying probiotics and prebiotics for human use. J. Nutr.2000, 130, 391–395. [Google Scholar]
- Montagne, L.; Pluske, J.R.; Hampson, D.J. A review of interactions between dietary fiber and the intestinal mucosa, and their consequences on digestive health in young non-ruminant animals. Anim. Feed Sci. Technol.2003, 108, 95–117. [Google Scholar] [CrossRef]
- Cook, S.I.; Sellin, J.H. Review article: Short chain fatty acids in health and disease. Aliment. Pharm. Therap.1998, 12, 499–507. [Google Scholar] [CrossRef]
- Lewis, S.; Heaton, K. Increasing butyrate concentration in the distal colon by accelerating intestinal transit. Gut1997, 41, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Sivieri, K.; Morales, M.L.V.; Saad, S.M.I.; Adorno, M.A.T.; Sakamoto, I.K.; Rossi, E.A. Prebiotic effect of fructooligosaccharide in the simulator of the human intestinal microbial ecosystem. J. Med. Food2014, 17, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, M.; Ushida, K.; Hoshi, S.; Kashima, N.; Ajisaka, K.; Yajima, T. Butyrate and propionate production from d-mannitol in the large intestine of pig and rat. Microb. Ecol. Health Dis.2005, 17, 169–176. [Google Scholar] [CrossRef]
- Xiao, J.; Li, X.; Min, X.; Sakaguchi, E. Mannitol improves absorption and retention of calcium and magnesium in growing rats. Nutrition2013, 29, 325–331. [Google Scholar] [CrossRef]
- Minamida, K.; Shiga, K.; Sujaya, I.N.; Sone, T.; Yokota, A.; Hara, H.; Asano, K.; Tomita, F. Effects of difructose anhydride III (DFA III) administration on rat intestinal microbiota. J. Biosci. Bioeng.2005, 99, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.; Gibson, G.R.; Delzenne, N. Biochemistry of oligofructose, a non-digestible fructooligosaccharide: An approach to estimate its caloric value. Nutr. Rev.1993, 51, 137–146. [Google Scholar] [CrossRef]
- Wang, X.; Gibson, G.R. Effect of the in vitro fermentation of oligofructose and inulin by bacteria growing in the human large intestine. J. Appl. Bacteriol.1993, 75, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol Lett.2009, 294, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Koleva, P.T.; Valcheva, R.S.; Sun, X.; Gänzle, M.G.; Dieleman, L.A. Inulin and fructo-oligosaccharides have divergent effects on colitis and commensal microbiota in HLA-B27 transgenic rats. Br. J. Nutr.2012, 108, 1633–1643. [Google Scholar] [CrossRef] [PubMed]
- McHan, F.; Shotts, E.B. Effect of short-chain fatty acids on the growth of Salmonella typhimurium in an in vitro system. Avian Dis.1993, 37, 396–398. [Google Scholar] [CrossRef] [PubMed]
- Rekiel, A.; Wiecek, J.; Bielecki, W.; Gajewska, J.; Cichowicz, M.; Kulisiewicz, J.; Batorska, M.; Roszkowski, T.; Beyga, K. Effect of addition of feed antibiotic flavomycin, or prebiotic BIO-MOS on production results of fatteners, blood biochemical parmeters, morphometric indices of intestine and composition of microflora. Arch. Tierz.2007, 50, 172–180. [Google Scholar]
- Koleva, P.; Ketabi, A.; Valcheva, R.; Gänzle, M.G.; Dieleman, L.A. Chemically defined diet alters the protective properties of fructo-oligosaccharides and isomalto-oligosaccharides in HLA-B27 transgenic rats. PLoS ONE2014, 9, e111717. [Google Scholar] [CrossRef] [PubMed]
- Gidenne, T.; Licois, D. Effect of high fiber intake on resistance of the growing rabbit to an experimental inoculation with an enteropathogenic strain of Escherichia coli. Anim. Sci.2005, 80, 281–288. [Google Scholar] [CrossRef]
- Mao, B.; Li, D.; Zhao, J.; Liu, X.; Gu, Z.; Chen, Y.Q.; Zhang, H.; Chen, W. Metagenomic insights into the effects of fructooligosaccharides (FOS) on the composition of fecal microbiota in mice. J. Agric. Food Chem.2015, 63, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Hütt, P.; Shchepetova, J.; Lõivukene, K.; Kullisaar, T.; Mikelsaar, M. Antagonistic activityof probiotic lactobacilli and bifidobacteria against entero- and uropathogens. J. Appl. Microbiol.2006, 100, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Gopal, P.K.; Sullivan, P.A.; Smart, J.B. Utilization of galacto-oligosaccharides as selective substrates for growth by lactic acid bacteria including Bifidobacterium lactis DR10 and Lactobacillus rhamnosus DR20. Int. Dairy J.2001, 11, 19–25. [Google Scholar] [CrossRef]
- Mäkeläinen, H.; Saarinen, M.; Stowell, J.; Rautonen, N.; Ouwehand, A.C. Xylo-oligosaccharides and lactitol promote the growth of Bifidobacterium lactis and Lactobacillus species in pure cultures. Benef. Microbes2010, 1, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Veerappan, G.R.; Betteridge, J.; Young, P.E. Probiotics for the treatment of inflammatory bowel disease. Curr. Gastroenterol. Rep.2012, 14, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Servin, A.L. Antagonistic activities of lactobacilli and bifidobacteria against microbial pathogens. Microbiol. Rev.2004, 28, 405–440. [Google Scholar] [CrossRef] [PubMed]
- Arora, T.; Sharma, R.K. Prebiotic effectiveness of galactooligosaccharides and β-glucan in stimulation of growth of Lactobacillus acidophilus NCDC 13 in vitro. Curr. Top. Nutraceutical Res.2011, 9, 67–70. [Google Scholar]
- Sangwan, V.; Tomar, S.K.; Ali, B.; Singh, R.R.B.; Singh, A.K. Hypoglycaemic effect of galactooligosaccharides in alloxan-induced diabetic rats. J. Dairy Res.2015, 82, 70–77. [Google Scholar] [CrossRef]
- Bach Knudsen, K.E. Effect of dietary non-digestible carbohydrates on the rate of SCFA delivery to peripheral tissues. Food Foods Ingredients J. Jpn.2005, 210, 1008–1017. [Google Scholar]
- Kien, C.L.; Blauwiekel, R.; Bunn, J.Y.; Jetton, T.L.; Frankel, W.L.; Holst, J.J. Cecal infusion of butyrate increases intestinal cell proliferation in piglets. J. Nutr.2007, 137, 916–922. [Google Scholar] [PubMed]
- Scheppach, W.; Dusel, G.; Kuhn, T.; Loges, C.; Karch, H.; Bartram, H.P. Effect of l-glutammine and n-butyrate o the restitution of rat colonic mucosa after acid induced injury. Gut1996, 38, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Lobo, A.R.; Gaievski, E.H.; de Carli, E.; Alvares, E.P.; Colli, C. Fructo-oligosaccharides and iron bioavailability inanaemic rats: The effects on iron species distribution, ferroportin-1expression, crypt bifurcation and crypt cell proliferation in the caecum. Br. J. Nutr.2014, 112, 1286–1295. [Google Scholar] [CrossRef]
- Blottière, H.M.; Buecher, B.; Galmiche, J.P.; Cherbut, C. Molecular analysis of the effect of short-chain fatty acids on intestinal cell proliferation. Proc. Nutr. Soc.2003, 62, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Henningsson, A.; Inger Björck, I.; Nyman, M. Short-chain fatty acid formation at fermentation of indigestible carbohydrates. Scand. J. Nutr.2001, 145, 165–168. [Google Scholar] [CrossRef]
- Mourão, J.L.; Pinheiro, V.; Alves, A.; Guedes, C.M.; Pinto, L.; Saavedra, M.J.; Spring, P.; Kocher, A. Effect of mannan oligosaccharides on the performance, intestinal morphology and cecal fermentation of fattening rabbits. Anim. Feed Sci. Technol.2006, 126, 107–120. [Google Scholar] [CrossRef]
- Guedes, C.M.; Mourão, J.L.; Silva, S.R.; Gomes, M.J.; Rodrigues, M.A.M.; Pinheiro, V. Effects of age and mannanoligosaccharides supplementation on production of volatile fatty acids in the caecum of rabbits. Anim. Feed Sci. Technol.2009, 150, 330–336. [Google Scholar] [CrossRef]
- Scheppach, W.; Weiler, F. The butyrate story: Old wine in new bottles? Curr. Opin. Clin. Nutr. Metab. Care2004, 7, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Nurmi, J.; Puolakkainen, P.; Rautonen, N. Bifidobacterium lactis sp. 420 up-regulates cylooxygenase [Cox] 1 and down-regulates Cox-2 gene expression in a Caco-2 cell culture model. Nutr. Can.2005, 51, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Berndt, B.E.; Zhang, M.; Owyang, S.Y.; Cole, T.S.; Wang, T.W.; Luther, J. Butyrate increases IL-23 production by stimulated dendritic cells. Am. J. Physiol. Gastrointest. Liver Physiol.2012, 303, 1384–1392. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, Y.; Obata, Y.; Fukuda, S.; Endo, T.A.; Nakato, G.; Takahashi, D. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature2013, 504, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Bohmig, G.A.; Krieger, P.M.; Saemann, M.D.; Wenhardt, C.; Pohanka, E.; Zlabinger, G.J. N-Butyrate down regulates the stimulatory function of peripheral blood derived antigen-presenting cells: A potential mechanism for modulating T-cell responses by short-chain fatty acids. Immunology1997, 92, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.V.; Hao, L.; Offermanns, S.; Medzhitov, R. The microbial metabolite butyrate regulates intestinal macrophage function via histone deacetylase inhibition. Immunology2014, 111, 2247–2252. [Google Scholar] [CrossRef] [PubMed]
- Komura, M.; Fukuta, T.; Genda, T.; Hino, S.; Aoe, S.; Kawagishi, H.; Tatsuya, M. A short-term ingestion of fructo-oligosaccharides increases immunoglobulin A and mucin concentrations in the rat cecum, but the effects are attenuated with the prolonged ingestion. Biosci. Biotechnol. Biochem.2014, 78, 1592–1602. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.Y.; Chio, Y.S. Dose-response assessment of the anti-cancer efficacy of soy isoflavones in dimethylhydrazine- treated rats fed 6% fructooligosaccharide. Nutr. Res. Pract.2008, 2, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Bielecka, M.; Biedrzycka, E.; Majkowska, A. Selection of probiotics and prebiotics for synbiotics and confirmation of their in vivo effectiveness. Food Res. Int.2002, 35, 139–144. [Google Scholar] [CrossRef]
- Pan, X.D.; Chen, F.Q.; Wu, T.X.; Tang, H.G.; Zhao, Z.Y. Prebiotic oligosaccharides change the concentrations of short-chain fatty acids and the microbial population of mouse bowel. J. Zhejiang Univ. Sci. B2009, 10, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Gudiel-Urbano, M.; Goñi, I. Effect of fructooligosaccharide on nutritional parameters and mineral bioavailability in rats. J. Sci. Food Agric.2002, 82, 913–917. [Google Scholar] [CrossRef]
- Xiao, J.; Okayama University, Okayama, Japan. Unpublished work. 2011.
- Silk, D.B.A.; Davis, A.; Vulevic, J.; Tzortzis, G.; Gibson, G.R. Clinical trial: The effects of a trans-galactooligosaccharide prebiotic on fecal microbiota and symptoms in irritable bowel syndrome. Aliment. Pharm. Therap.2009, 29, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zeng, T.; Wang, S.E.; Li, F.; Guo, X.Y.; Jin, J.; Yu, H.X. Laxative effect of fructooligosaccharide in mice and humans. Curr. Top. Nutraceutical Res.2013, 11, 1–8. [Google Scholar]
- Costalos, C.; Kapiki, A.; Apostolou, M.; Papathoma, E. The effect of a prebiotic supplemented formula on growth and stool microbiology of term infants. Early Hum. Dev.2008, 84, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Mihatsch, W.; Hoegel, J.; Pohlandt, F. Prebiotic oligosaccharides reduce stool viscosity and accelerate gastrointestinal transport in preterm infants. Acta Paediatr.2006, 95, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Tateyama, I.; Hashii, K.; Johno, I.; Iino, T.; Hirai, K.; Suwa, Y.; Kiso, Y. Effect of xylooligosaccharide intake on severe constipation in pregnant women. J. Nutr. Sci. Vitaminol.2005, 51, 445–448. [Google Scholar] [CrossRef] [PubMed]
- Pansu, D.; Chapuy, M.C.; Milani, M.; Bellaton, C. Transepithelial calcium transport enhanced by xylose and glucose in the rat jejunal ligated loop. Calcified Tissue Int.1976, 21, 45–52. [Google Scholar] [CrossRef]
- Cummings, J.H. The Large Intestine in Nutrition and Disease; Institut Danone: Brussels, Belgium, 1997; pp. 1–155. [Google Scholar]
- Ruppin, H.; Bar-Meir, S.; Soergel, K.H.; Wood, C.M.; Schmitt, M.G. Absorption of short chain fatty acids by the colon. Gastroenterology1980, 78, 1500–1507. [Google Scholar] [PubMed]
- Freitas Kde, C.; Amancio, O.M.; de Morais, M.B. High-performance inulin and oligofructose prebiotics increase the intestinal absorption of iron in rats with iron deficiency anaemia during the growth phase. Br. J. Nutr.2012, 108, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Nakada, H.; Sakae, T.; Watanabe, T.; Takahashi, T.; Fujita, K.; Tanimoto, Y. A new osteoporosis prevention supplements-diet improve bone mineral density in ovariectomized rats. J. Hard Tissue Biol.2014, 23, 1–8. [Google Scholar] [CrossRef]
- Lobo, A.R.; Colli, C.; Filisetti, T.M.C.C. Fructooligosaccharides improve bone mass and biomechamical properties in rats. Nutr. Res.2006, 26, 413–420. [Google Scholar] [CrossRef]
- Takasugi, S.; Ashida, K.; Maruyama, S.; Matsukiyo, Y.; Kaneko, T.; Yamaji, T. A combination of a dairy product fermented by lactobacilli and galactooligosaccharides shows additive effects on mineral balances in growing rats with hypochlorhydria induced by a proton pump inhibitor. Biol. Trace Elem. Res.2013, 153, 309–318. [Google Scholar] [CrossRef]
- Zafar, T.A.; Weaver, C.M.; Zhao, Y.D.; Martin, B.R.; Wastney, M.E. Nondigestible oligosaccharides increase calcium absorption and suppress bone resorption in ovariectomized rats. J. Nutr.2004, 134, 399–402. [Google Scholar]
- Heaney, R.P. Nutrition and risk for osteoporosis. In Osteoporosis, 1st ed.; Marcus, R., Feldman, D., Eds.; Academic Press: San Diego, CA, USA, 1996; pp. 483–509. [Google Scholar]
- Kishino, E.; Norii, M.; Fujita, K.; Hara, K.; Teramoto, F.; Fukunaga, M. Enhancement by lactosucrose of the calcium absorption from the intestine in growing rats. Biosci. Biotechnol. Biochem.2006, 70, 1485–1488. [Google Scholar] [CrossRef]
- Metzler-Zebeli, B.U.; Deckardt, K.; Schollenberger, M.; Rodehutscord, M.; Zebeli, Q. Lactic acid and thermal treatments trigger the hydrolysis of myo-inositol hexakisphosphate and modify the abundance of lower myo-inositol phosphates in barley (Hordeum vulgare L.). PLoS ONE2014, 9, e101166. [Google Scholar] [CrossRef]
- Weaver, C.M.; Martin, B.R.; Nakatsu, C.H. Galactooligosaccharides improve mineral absorption and bone properties in growing rats through gut fermentation. J. Agric. Food Chem.2011, 59, 6501–6510. [Google Scholar] [CrossRef]
- Takasugi, S.; Ashida, K.; Maruyama, S.; Komaba, Y.; Kaneko, T.; Yamaji, T. A dairy product fermented by lactobacilli cancels the adverse effects of hypochlorhydria induced by a proton pump inhibitor on bone metabolism in growing rats. Br. J. Nutr.2011, 106, 1487–1494. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zeng, T.; Wang, S.E.; Wang, W.; Wang, Q.; Yu, H.X. Fructo-oligosaccharides enhance the mineral absorption and counteract the adverse effects of phytic acid in mice. Nutrition2010, 26, 305–311. [Google Scholar]
- Scholz-Ahrens, K.E.; Schrezenmeir, J. Inulin and oligofructose and mineral metabolism: The evidence from animal trials. J. Nutr.2007, 137, 2513–2523. [Google Scholar]
- Johnson, C.D.; Lucas, E.A.; Hooshmand, S.; Campbell, S.; Akhter, M.P.; Arjmandi, B.H. Addition of fructooligosaccharides and dried plum to soy-based diets reverses bone loss in the ovariectomized rat. Evid. Based Complement. Alternat. Med.2011. [Google Scholar] [CrossRef]
- Sato, H.; Ide, Y.; Nasu, M. The effects of oral xylitol administration on bone density in rat femur. Odontology2011, 99, 28–33. [Google Scholar] [CrossRef]
- Hamalainen, M.M. Retention of calcium from various xylitolcalcium combinations in rats. Proc. Soc. Exp. Biol. Med.1994, 205, 253–256. [Google Scholar] [CrossRef]
- Goda, T.; Suruga, K.; Takase, S.; Ezawa, I.; Hosoya, N. Dietary maltitol increases calcium content and breaking force of femoral bone in ovariectomized rats. J. Nutr.1995, 125, 2869–2873. [Google Scholar] [PubMed]
- Mattila, P.T.; Svanberg, M.J.; Jämsä, T.; Knuuttila, M.L. Improved bone biomechanical propertiesin xylitol-fed aged rats. Metabolism2002, 51, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Goda, T.; Kishi, K.; Ezawa, I.; Takase, S. The maltitol-induced increase in intestinal calcium transport increases the calcium content and breaking force of femoral bone in weanling rats. J. Nutr.1998, 128, 2028–2031. [Google Scholar] [PubMed]
- Kaivosoja, S.M.; Mattila, P.T.; Knuuttila, M.L. Dietary xylitol protects against the imbalance in bone metabolism during the early phase of collagen type II-induced arthritis in dark agouti rats. Metabolism2008, 57, 1052–1055. [Google Scholar] [CrossRef] [PubMed]
- Younes, H.; Demigne, C.; Behr, S.R.; Garleh, K.A.; Remesy, C. A blend of dietary fibers increases urea disposal in the large intestine and lowers urinary nitrogen excretion in rats fed a low protein diet. J. Nutr. Biochem.1996, 7, 474–480. [Google Scholar] [CrossRef]
- Hara, H.; Onoshima, S.; Nakagawa, C. Difructose anhydride III promotes iron absorption in the large intestine. Nutrition2010, 26, 120–127. [Google Scholar] [CrossRef]
- Karbach, U.; Feldmeier, H. The cecum is the site with the highest calcium absorption in rat intestine. Dig. Dis. Sci.1993, 38, 1815–1824. [Google Scholar] [CrossRef]
- Mineo, H.; Hara, H.; Tomita, F. Short-chain fatty acids enhance diffusional Ca transport in the epithelium of the rat cecum and colon. Life Sci.2001, 69, 517–526. [Google Scholar] [CrossRef]
- Scholz-Ahrens, K.E.; Açil, Y.; Schrezenmeir, J. Effect of oligofructose or dietary calcium on repeated calcium and phosphorus balances, bone mineralization and trabecular structure in ovariectomized rats. Br. J. Nutr.2002, 88, 365–377. [Google Scholar] [CrossRef]
- Scholz-Ahrens, K.E.; Schrezenmeir, J. Inulin, oligofructose and mineral metabolism—Experimentaldata and mechanism. Br. J. Nutr.2002, 87, 179–186. [Google Scholar] [CrossRef]
- Younes, H.; Coudray, C.; Bellanger, J.; Demigné, C.; Rayssiguier, Y.; Rémésy, C. Effects of two fermentable carbohydrates (inulin and resistant starch) and their combination on calcium and magnesium balance in rats. Br. J. Nutr.2001, 86, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Raschka, L.; Daniel, H. Mechanisms underlying the effects of inulin-type fructans on calcium absorption in the large intestine of rats. Bone2005, 37, 728–735. [Google Scholar] [CrossRef]
- Chonan, O.; Matsumoto, K.; Watanuki, M. Effect of galactooligosaccharides on calcium absorption and preventing bone loss in ovariectomized rats. Biosci. Biotechnol. Biochem.1995, 59, 236–239. [Google Scholar] [CrossRef]
- Samanta, A.K.; Jayapal, N.; Senani, S.; Kolte, A.P.; Sridhar, M. Prebiotic inulin: Useful dietary adjuncts to manipulate the livestock gut microflora. Braz. J. Microbiol.2013, 44, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Topping, D.L.; Clifton, P.M. Short-chain fatty acids and human colonic function: Roles of resistant starch and nonstarch polysaccharides. Physiol. Rev.2001, 81, 1031–1064. [Google Scholar]
- Lutz, T.; Scharrer, E. Effect of short-chain fatty acids on calcium absorption by the rat colon. Exp. Physiol.1991, 76, 615–618. [Google Scholar] [CrossRef]
- Asvarujanon, P.; Ishizuka, S.; Hara, H. Promotive effects of non-digestible disaccharides on rat mineral absorption depend on the type of saccharide. Nutrition2005, 21, 1025–1035. [Google Scholar] [CrossRef]
- Ohta, A.; Sakai, K.; Takasaki, M.; Uehara, M.; Tokunaga, T.; Adachi, T. Dietary heme iron does not prevent postgastrectomy anemia but fructooligosaccharides improve bioavailability of heme iron in rats. Int. J. Vitam. Nutr. Res.1999, 69, 348–355. [Google Scholar] [CrossRef]
- Ohta, A.; Baba, S.; Takizawa, T.; Adachi, T. Effects of fructooligosaccharides on the absorption of magnesium in the magnesium-deficient rat model. J. Nutr. Sci. Vitaminol.1994, 40, 171–181. [Google Scholar] [CrossRef]
- Ohta, A.; Ohtsuki, M.; Baba, S.; Takizawa, T.; Adachi, T.; Kimura, S. Effects of fructooligosaccharides on the absorption of iron, calcium and magnesium in iron-deficient anemic rats. J. Nutr. Sci. Vitaminol.1995, 41, 281–291. [Google Scholar] [CrossRef]
- Shiga, K.; Nishimukai, M.; Tomita, F.; Hara, H. Ingestion of difructose anhydride III, a non-digestible disaccharide, prevents gastrectomy-induced iron malabsorption and anemia in rats. Nutrition2006, 22, 786–793. [Google Scholar] [CrossRef]
- Suzuki, T.; Nishimukai, M.; Shinoki, A.; Taguchi, H.; Fukiya, S.; Yokota, A.; Saburi, W.; Yamamoto, T.; Hara, H.; Matsui, H. Ingestion of epilactose, a non-digestible disaccharide, improves postgastrectomy osteopenia and anemia in rats through the promotion of intestinal calcium and iron absorption. J. Agric. Food Chem.2010, 58, 10787–10792. [Google Scholar] [CrossRef]
- Santos, E.F.D.; Tusboi, K.H.; Araújo, M.R.; Falconi, M.A.; Ouwehand, A.C.; Andreollo, N.A.; Miyasaka, C.K. Ingestion of polydextrose increase the iron absorption in rats submitted to partial gastrectomy. Acta Cir. Bras.2010, 25, 518–524. [Google Scholar] [CrossRef]
- Santos, E.F.D.; Tusboi, K.H.; Araújo, M.R.; Andreollo, N.A.; Miyasaka, C.K. Dietary galactooligosaccharides increase calcium absorption in normal and gastrectomized rats. Rev. Col. Bras. Cir.2011, 38, 186–190. [Google Scholar] [CrossRef]
- Mitamura, R.; Hara, H. Prolonged feeding of difructose anhydride III increases strength and mineral concentrations of the femur in ovariectomized rats. Br. J. Nutr.2005, 94, 268–274. [Google Scholar] [CrossRef]
- Weisstaub, A.R.; Victoria Abdala, V.; Chaves, M.G.; Mandalunis, P.; Zuleta, Á.; Zeni, S. Polydextrose enhances calcium absorption and bone retention in ovariectomized rats. Int. J. Food Sci.2013. [Google Scholar] [CrossRef]
- Ohta, A.; Ohtuki, M.; Takizawa, T.; Inaba, H.; Adachi, T.; Kimura, S. Effects of fructooligosaccharides on the absorption of magnesium and calcium by cecectomized rats. Int. J. Vitam. Nutr. Res.1994, 64, 316–323. [Google Scholar]
- Heijnen, A.M.; Brink, E.J.; Lemmens, A.G.; Beynen, A.C. Ileal pH and apparent absorption of magnesium in rats fed on diets containing either lactose or lactulose. Br. J. Nutr.1993, 70, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Fukahori, M.; Sakurai, H.; Akatsu, S.; Negishi, M.; Sato, H.; Goda, T.; Takase, S. Enhanced absorption of calcium after oral administration of maltitol in the rat intestine. J. Pharm. Pharmacol.1998, 50, 1227–1232. [Google Scholar] [CrossRef] [PubMed]
- Mineo, H.; Hara, H.; Shigematsu, N.; Okuhara, Y.; Tomita, F. Melibiose, difructose anhydride III and difructose anhydride IV enhance net calcium absorption in rat small and large intestinal epithelium by increasing the passage of tight junctions in vitro. J. Nutr.2002, 132, 3394–3399. [Google Scholar] [PubMed]
- Mineo, H.; Amamo, M.; Minaminida, K.; Chiji, H.; Shigematsu, N.; Tomita, F.; Hara, H. Two-week feeding of difructose anhydride III enhances calcium absorptive activity with epithelial cell proliferation in isolated rat cecal mucosa. Basic Nutr. Investig.2006, 22, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Min, X.; Tsuzuki, Y.; Sakaguchi, E. Effect of indigestible sugars on nitrogen utilization in adult rabbits. Anim. Sci. J.2011, 82, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Min, X.; Li, X.; Hiura, S.; Kawasaki, K.; Xiao, J.; Sakaguchi, E. Effect of d-mannitol on nitrogen retention, fiber digestibility and digesta transit time in adult rabbits. Anim. Sci. J.2013, 84, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, A.; Nishioka, S.; Islam, S.M.; Sakaguchi, E. Mannitol lowers fat digestibility and body fat accumulation in both normal and cecectomized rats. J. Nutr. Sci. Vitaminol.2009, 55, 242–251. [Google Scholar] [CrossRef]
- Sakaguchi, E.; Sakoda, C.; Toramaru, Y. Caecal fermentation and energy accumulation in the rat fed on indigestible oligosaccharides. Br. J. Nutr.1998, 80, 469–476. [Google Scholar]
- Younes, H.; Garleb, K.; Behr, S.; Rémésy, C.; Demigné, C. Fermentable fibers or oligosaccharides reduce urinary nitrogen excretion by increasing urea disposal in the rat cecum. J. Nutr.1995, 125, 1010–1016. [Google Scholar]
- Bovera, F.; Lestingi, A.; Marono, S.; Iannaccone, F.; Nizza, S.; Mallardo, K.; de Martino, L.; Tateo, A. Effect of dietary mannan-oligosaccharides on in vivo performance, nutrient digestibility and caecal content characteristics of growing rabbits. J. Anim. Physiol. Anim. Nutr.2012, 96, 130–136. [Google Scholar] [CrossRef]
- Carabaño, R.; Piquer, J. The digestive system of the rabbit. In The Nutrition of the Rabbit; De Blas, C., Wiseman, J., Eds.; CAB International: Wallingford, CT, USA, 1998; pp. 1–16. [Google Scholar]
- Kawasaki, K.; Min, X.; Li, X.; Hasegawa, E.; Sakaguchi, E. Transfer of blood urea nitrogen to cecal microbial nitrogen is increased by fructo-oligosaccharide feeding in guinea pigs. Anim. Sci. J.2015, 86, 77–82. [Google Scholar] [CrossRef]
- Xiao, L.; Xiao, M.; Jin, X.; Kawasaki, K.; Ohta, N.; Sakaguchi, E. Transfer of blood urea nitrogen to cecal microbial nitrogen is increased by mannitol feeding in growing rabbits fed timothy hay diet. Animal2012, 6, 1757–1763. [Google Scholar] [CrossRef]
- Belenguer, A.; Balcells, J.; Guada, J.A.; Decoux, M.; Milne, E. Protein recycling in growing rabbits: Contribution of microbial lysine to amino acid metabolism. Br. J. Nutr.2005, 94, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Hanieh, H.; Sakaguchi, E. Effect of d-mannitol on feed digestion and cecotrophic system in rabbits. Anim. Sci. J.2009, 80, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Min, X.; Xiao, J.; Kawasaki, K.; Ohta, N.; Sakaguchi, E. Utilization of dietary urea nitrogen is stimulated by d-mannitol feeding in rabbits. Anim. Sci. J.2012, 83, 605–609. [Google Scholar] [CrossRef]
- Yamada, K.; Hidaka, H.; Inooka, G.; Iwamoto, Y.; Kuzuya, T. Plasma fructosemic and glycosemic responses to fructooligosaccharides in rats and healthy human subjects. Dig. Absorpt.1990, 13, 88–91. [Google Scholar]
- Kaume, L.; Gilbert, W.; Gadang, V.; Devareddy, L. Dietary supplementation of fructooligosaccharides reduces hepatic steatosis associated with insulin resistance in obese zucker rats. Funct. Foods Heaths Dis.2011, 5, 199–213. [Google Scholar]
- Costa, G.T.; Guimarães, S.B.; de Carvalho Sampaio, H.A. Fructo-oligosaccharide effects on blood glucose: An overview. Acta Cir. Bras.2012, 27, 279–282. [Google Scholar] [CrossRef]
- Mäkinen, K.K.; Hämäläinem, M.M. Metabolic effects in rats of high oral doses of galactitol, mannitol and xylitol. J. Nutr.1985, 115, 890–899. [Google Scholar]
- Respondek, F.; Hilpipre, C.; Chauveau, P.; Cazaubiel, M.; Gendre, D.; Maudet, C.; Wagner, A. Digestive tolerance and postprandial glycaemic and insulinaemic responses after consumption of dairy desserts containing maltitol and fructo-oligosaccharides in adults. Eur. J. Clin. Nutr.2014, 68, 575–580. [Google Scholar] [CrossRef]
- Ellwood, K.C.; Bhathena, S.J.; Johannessen, J.N.; Bryane, M.A.; O’Donnell, M.W. Biomarkers used to assess the effect of dietary xylitol or sorbitol in the rat. Nutr. Res.1999, 19, 1637–1648. [Google Scholar] [CrossRef]
- Urushima, H.; Sanada, Y.; Sakaue, M.; Matsuzawa, Y.; Ito, T.; Maeda, K. Maltitol prevents the progression of fatty liver degeneration in mice fed high-fat diets. J. Med. Food2015. [Google Scholar] [CrossRef] [PubMed]
- Nishimukai, M.; Watanabe, J.; Taguchi, H.; Senoura, T.; Hamada, S.; Matsui, H.; Yamamoto, T.; Wasaki, J.; Hara, H.; Ito, S. Effects of epilactose on calcium absorption and serum lipid metabolism in rats. J. Agric. Food Chem.2008, 56, 10340–10345. [Google Scholar] [CrossRef]
- Costa, G.T.; de Abreu, G.C.; Guimarães, A.B.B.; de Vasconcelos, P.R.L.; Guimarães, S.B. Fructo-oligosaccharide effects on serum cholesterol levels. An overview. Acta Cir. Bras.2015, 30, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Merino-Aguilar, H.; Arrieta-Baez, D.; Jiménez-Estrada, M.; Magos-Guerrero, G.; Hernández-Bautista, R.J.; Alarcón-Aguilar, F.J. Effect of fructooligosaccharides fraction from Psacalium decompositum on inflammation and dyslipidemia in rats with fructose-induced obesity. Nutrients2014, 6, 591–604. [Google Scholar] [CrossRef] [PubMed]
- Begley, M.; Hill, C.; Gahan, C.G.M. Bile salt hydrolase activity in probiotics. Appl. Environ. Microbiol.2006, 72, 1729–1738. [Google Scholar] [CrossRef]
- Taylor, G.R.; Williams, C.M. Effects of probiotics and prebiotics on blood lipids. Br. J. Nutr.1998, 80, 225–230. [Google Scholar]
- Incoll, L.; Bonnett, G.D. The occurrence of fructan in food plants. In Inulin and Inulin Containing Crops; Fuchs, A., Ed.; Elsevier Science Publishers BV: Amsterdam, The Netherlands, 1993; pp. 309–319. [Google Scholar]
- Daubioul, C.A.; Rousseau, N.; Demeure, R.; Gallez, B.; Taper, H.; Declerck, B.; Delzenne, N.M. Dietary fructans, but not cellulose, decrease triglyceride accumulation in the liver of obese Zucker fa/fa rats. J. Nutr.2002, 132, 967–973. [Google Scholar]
- Beylot, M. Effects of inulin-type fructans on lipid metabolism in man and in animal models. Br. J. Nutr.2005, 93, 163–168. [Google Scholar] [CrossRef]
- Chen, H.; Liu, L.J.; Zhu, J.J.; Xu, B.; Li, R. Effect of soybean oligosaccharides on blood lipid, glucose levels and antioxidant enzymes activity in high fat rats. Food Chem.2010, 119, 1633–1636. [Google Scholar] [CrossRef]
- Li, X.J.; Piao, X.S.; Kim, S.W.; Liu, P.; Wang, L.; Shen, Y.B.; Jung, S.C.; Lee, H.S. Effects of chito-oligosaccharide supplementation on performance, nutrient digestibility, and serum composition in broiler chickens. Poultry Sci.2007, 86, 1107–1114. [Google Scholar] [CrossRef]
- Kim, M.H.; Shin, H.K. The water-soluble extract of chicory influences serum and liver lipid concentrations, cecal short-chain fatty acid concentrations and fecal lipid excretion in rats. J. Nutr.1998, 128, 1731–1736. [Google Scholar]
- El-Mahmoudy, A.M.; Abdel-Fattah, F.A.; Abd El-Mageid, A.D.; Gheith, I.M. Effect of the growth promotant mannan-oligosaccharide on the lipogram and organ function profile in hyperlipidemic albino rats. Am. J. Phytomed. Clin. Ther.2014, 2, 334–347. [Google Scholar]






